The important things we should know about sterile vs non sterile gloves are their qualifications and designated purposes in certain duties.
Sterile gloves definition provided by certifications of the U.S. Food and Drug Administration (FDA) for their techniques. They are formulated to perform surgical medical procedures. Meanwhile, non sterile gloves are for medical examinations and other non-surgical services.
Regarding the Acceptable quality level (AQL) of pinholes, sterile gloves have to meet a stricter requirement of below 1.5% per tested set, while non sterile gloves’ AQL is below 2.5 %.
Table of Contents
FDA Medical Device Guide About Sterile Gloves
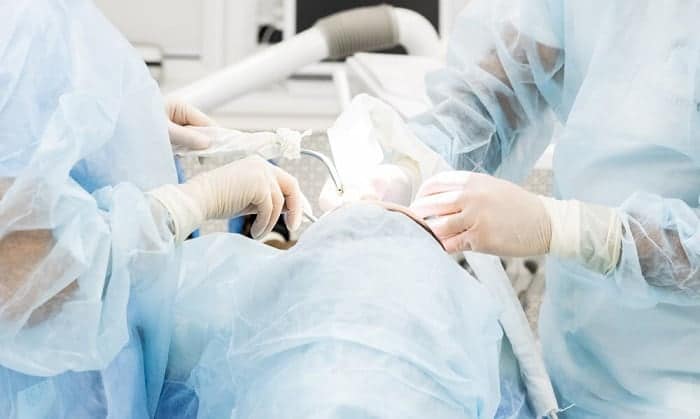
According to FDA standards, the sterile gloves of surgeons must be made from synthetic rubber. As surgical procedures require medical personnel to protect the patient’s wounds from any possible contaminations and surgical site infections, their device, including gloves, must be sterile.
Please note that this requirement does not include the dusting powder or lubricating agent applied to the sterile gloves.
Regarding the classifications of FDA standards for sterilization techniques of surgical gloves, it is the Class 1 device. The sterile gloves meet the design requirements of the FDA Quality System Regulation for notifying 90 days prior to the marketing process.
When producing sterile gloves, manufacturers must have their products undergo a sterilization process to be tested whether they meet the FDA’s Standard Assurance Level (SAL).
In detail, the sterility test of the sterile technique is based on the requirement of a SAL of 10^-6. And it requires a SAL of 10^− 3 for higher risk procedures of intact skin contact in surgery.
Here’re the sterilization standards recognized by the FDA:
- ANSI/AAMI/ISO 11135: Ethylene oxide sterilization validation and control
- AAMI/CDV 11137: Health care product and radiation sterilization
- AAMI/ISO: Health care product sterilization and radiation sterilization technical information report
A sample set of sterile gloves must meet the acceptable quality level (AQL) to have pinholes of 1.0 – 1.5 %. Also, surgical gloves must meet the performance criteria of biocompatibility in medical procedures and resistance to tears and leaks.
FDA Medical Device Guide About Non Sterile Gloves
Non sterile meaning the glove does not conform to the sterility assurance level (SAL). For non surgical procedures like examining in dental procedures, a non sterile glove suffices. Common single-used devices for medical care are poly gloves (latex free for medical personnel or patients with latex allergy), vinyl gloves (powder and latex free), and nitrile pairs (powder and natural rubber latex free).
There are some standards for this type of gloves, including:
- ADA Spec No. 102: Dentistry non sterile nitrile gloves
- CAN 20.27-M91: Non-sterile and sterile medical examination gloves for single-use
Regarding the acceptable quality level of non sterile gloves in a sample set, its AQL must be from 1.5 – 2.5. That means the number of gloves with pinholes in the submitted gloves for testing must be below 2.5%.
Hand Hygiene and other Requirements in Using Sterile Gloves vs Non Sterile Gloves
- Follow proper washing techniques for your hands before wearing gloves to perform medical services. The process must involve antimicrobial soap, hot water, and clean towels.
- Conform to the hand hygiene practice (washing hands) before and after examining or performing any medical service on an individual.
- Ensure a good fit of the gloves on your hands before handling patient care
- Use powder-free and latex-free gloves for allergic patients
- Be aware of pinholes in gloves, both from manufacturing and sharp devices or tools around.
- Change the gloves in case of ripping and tearing.
- Must not reuse sterile and non sterile gloves for medical care
- Must not disinfect and wash medical gloves by yourself for reuse
- Must not share gloves with other people
Conclusion
There must be no haphazard behaviors in using medical gloves, so everyone should be aware of certain information. For instance, we must know the difference between sterile vs non-sterile gloves and follow the regulation for their utilization.
Also, to purchase the right type of device for our work, make sure to check out the qualifications and standards that the gloves must meet. That way, we can protect our health and other’s safety while handling daily duties.
Do not hesitate to reach out to us in case you have any confusion about this guide.

Hi, I am Alexander, forty-seven years old this year. I have been in the construction industry for twenty-five years and have provided a wide range of services. When it comes to expert guidance on site safety gear, you can put your trust in me.